How Hexa-Aqua Coordination Compares to Basic Hydration States
When minerals dissolve in water, they do not exist as isolated particles. Instead, surrounding water molecules organize around the charged ion. Chemists often describe several conceptual hydration states.
 Fig. 1 Bare Ions
Fig. 1 Bare Ions
Theoretical Unhydrated Metal Ions
A bare ion refers to a metal ion without surrounding water molecules. In practice, bare ions rarely exist in water because the electric charge of the ion immediately attracts nearby water molecules. Bare ions are therefore mainly used as a theoretical reference to help scientists understand how hydration begins.[1]
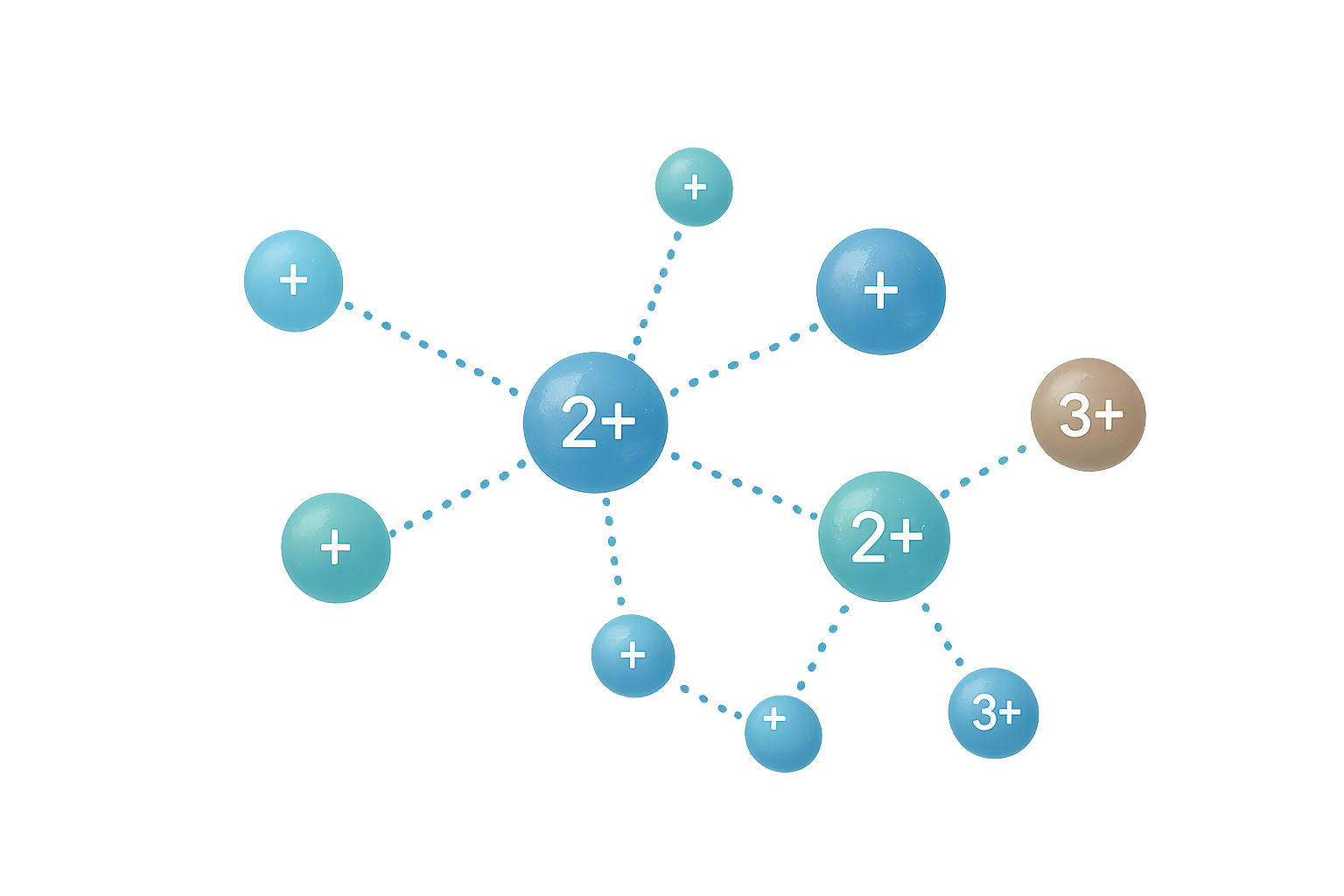 Fig. 2 Dynamic Hydration Shells
Fig. 2 Dynamic Hydration Shells
Rapidly Exchanging Water Layers
When a mineral dissolves in water, it becomes surrounded by a hydration shell. In many environments this shell is dynamic, meaning water molecules continuously exchange in and out. These rapidly exchanging shells allow mineral ions to:
- interact with proteins
- participate in enzyme reactions
- bind to biological molecules
Because the surrounding water molecules are constantly changing, the ion remains chemically active and able to interact with its environment.
Hexa-Aqua Hydration Shell
In many aqueous systems, metal ions form a coordination structure in which six water molecules surround the central ion in a stable arrangement: M(H₂O)₆²⁺.
The oxygen atoms coordinate to the central metal ion, forming a structured first hydration shell. Compared with loosely organized layers, this more structured environment can influence:
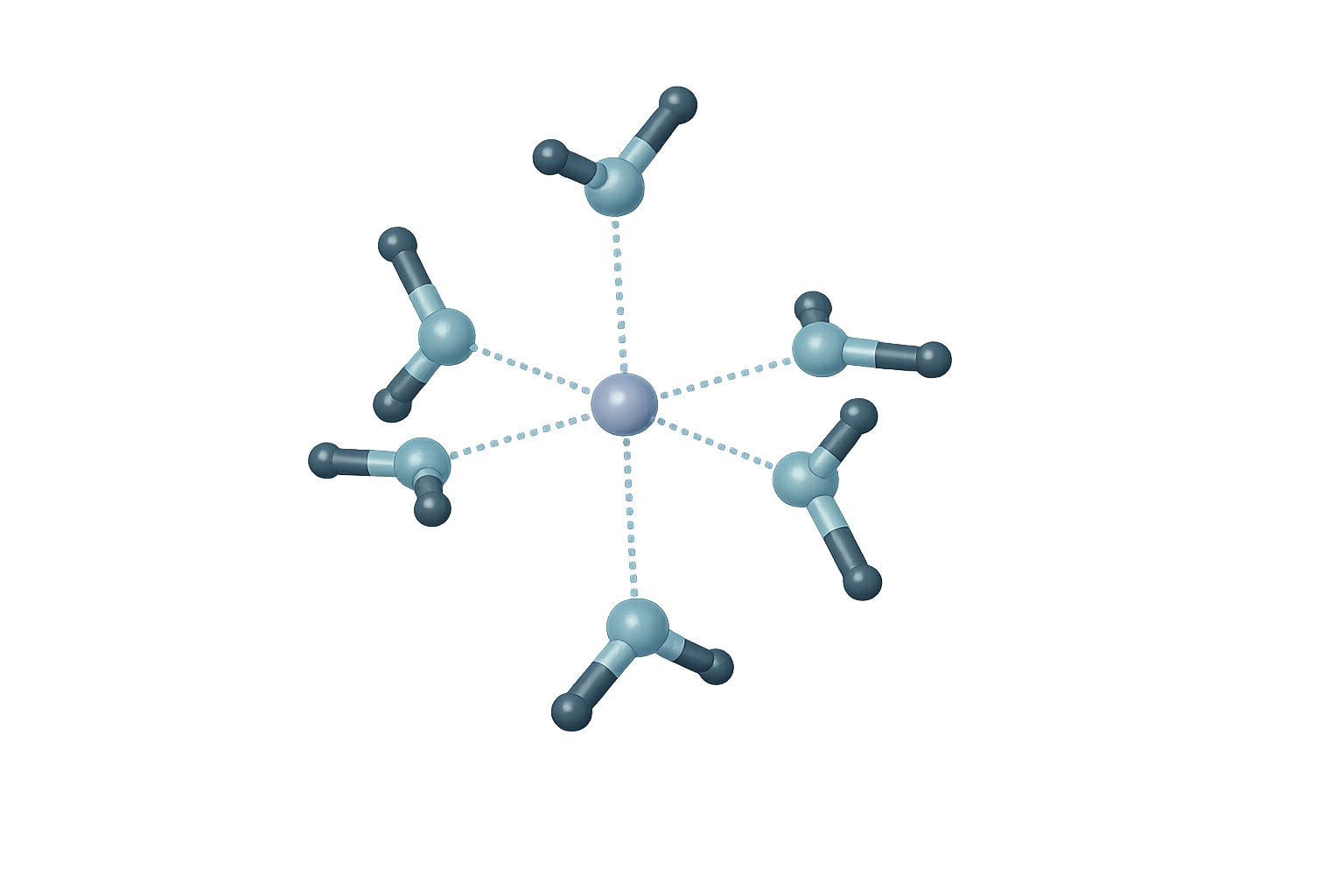 Fig. 3 Hexa-Aqua Coordination
Fig. 3 Hexa-Aqua Coordination
Hydration structure is an important factor in understanding how mineral ions behave in aqueous biological systems.[2]
Essential Minerals in the Body
Magnesium (Mg²⁺)
Magnesium ions maintain one of the more strongly bound hydration shells among biologically important metal ions. This hydration structure influences how magnesium interacts with ATP and many enzymes involved in cellular energy metabolism.[3]
Magnesium supports hundreds of enzyme systems and plays a central role in normal cellular energy metabolism.[4]
Zinc (Zn²⁺)
Zinc ions in water are initially surrounded by water molecules but readily exchange these molecules when interacting with proteins. This flexibility allows zinc to serve as both a structural and catalytic component.[5]
Zinc also participates in cellular signaling processes sometimes referred to as zinc signaling.[6]
Copper (Cu²⁺)
Copper is involved in mitochondrial energy production and antioxidant defense systems. Within cells, copper is tightly controlled by specialized transport and chaperone proteins.[7]
Copper balance in the body is maintained through complex transport systems that regulate its movement.[8]
Hexa-Aqua Coordination and Cellular Biochemistry
Coordination chemistry influences how mineral ions interact with enzymes, proteins, membranes, and metabolic pathways. Hydration structure, water exchange rates, and hexa-aqua geometry all contribute to how magnesium, zinc, and copper function in biological systems. Understanding these coordination environments provides important insight into how minerals behave in aqueous biological systems.
Selected References
[1] Ohtaki H., Radnai T. Structure and dynamics of hydrated ions. Chemical Reviews. 1993.
[2] Helm L., Merbach A. Inorganic and Bioinorganic Solvent Exchange Mechanisms. Chemical Reviews. 2005.
[3] Bleuzen A. et al. Water exchange on magnesium ions. Journal of the American Chemical Society. 1997.
[5] Vallee B.L., Auld D.S. Zinc coordination, function, and structure. Biochemistry. 1990.
[6] Maret W. Zinc Signals in Cellular Processes. Metallomics. 2017.
[8] Lutsenko S. Human copper homeostasis. Annual Review of Nutrition.
